Till and McCulloch worked out a system for measuring the radiation sensitivity of bone marrow cells. The researchers accomplished this feat by zapping mice with a dose that would kill the animals within 30 days if they did not receive a bone marrow transplant of fresh, undamaged cells. To obtain the donor material, the team divided bone marrow from unirradiated animals into portions, and exposed each to a different amount of radiation. The largest dose killed enough donor cells to obliterate their ability to rescue the mice; the smallest dose left much of it intact. The investigators knew how many unirradiated cells were needed to save the animals, so by counting the mouse survivors, they could infer the number of cells that had withstood a given amount of radiation.
Clumps or clones?
The scientists subsequently repeated the experiment but performed autopsies on the animals 10 days after transplantation. They noticed spleen nodules that contained dividing cells, some of which were specializing — or differentiating — into the three main types of blood cells: red cells, white cells, and platelets. The number of nodules was directly proportional to the number of live marrow cells the irradiated animals had received. The crucial entity was rare: About 10,000 marrow cells had to be injected for each nodule observed.
Aspects of the experiment and its results reminded the researchers of the test for live bacteria, which depends on the ability to reproduce. Scientists disperse bacteria on a Petri dish and each bacterium multiplies to form a colony. Counting colonies reveals the number of viable cells that were in the original sample. McCulloch and Till’s experiment, however, didn’t distinguish whether the spleen nodules originated from single cells that reproduced and differentiated, or came from clumps of multiple kinds of cells that then simply divided. The researchers wanted to find out whether all of the cells in a nodule — or colony, adopting the language of bacteriology — descended from a single cell (and thus represented a clone) or from multiple cells.
To accomplish this task, they needed cells that carried unique inheritable markers. They realized that irradiating cells would produce — at low frequency — exactly such markers in the form of visibly abnormal chromosomes. By dissecting spleen nodules into their constituent cells, Till and McCulloch could determine whether each cell from a given nodule contained the same rare chromosome. If so, the ‘colony-forming unit’ must have been a single cell; if not, it must have been composed of multiple cells.
Andrew Becker, a graduate student working with McCulloch and Till, examined hundreds of cells from 42 nodules obtained from 36 animals. Most contained only normal cells but four contained cells with distinctive chromosomes. Almost all of the dividing cells in each of these nodules carried a unique chromosomal alteration. The colonies thus arose from a single cell.
Till and McCulloch next wanted to know whether the colony-forming cells could renew themselves, forming new colony-forming cells. To answer this question, they and their colleague Louis Siminovitch broke up spleen nodules into their cellular components. The scientists then injected irradiated mice such that each animal received most of the cells from a single colony. If the colony-forming cells could duplicate themselves, the second-round animals would develop nodules. They did, thus establishing that colony-forming cells can self renew.
Later, the team showed that the multiple cell types within a colony arose from a single cell. This experiment addressed a key question in hematology at the time — whether three separate types of precursor cells headed the lineages that produced red cells, white cells, and platelets, or whether a single common stem cell gave rise to all three lineages.
By the early 1970s, Till and McCulloch’s experimental observations were clear-cut: They revealed that bone marrow transplantation owes its restorative powers to a single type of cell that not only can divide, but can differentiate into all three types of mature blood cells — red cells, white cells, and platelets. These features meant that the colony-forming cells represented a new class of progenitor cells — ones that could proliferate enough to repopulate the bone marrow of an entire animal, self-renew, and give rise to specialized cells that have limited life spans. This definition of a stem cell still holds true today.
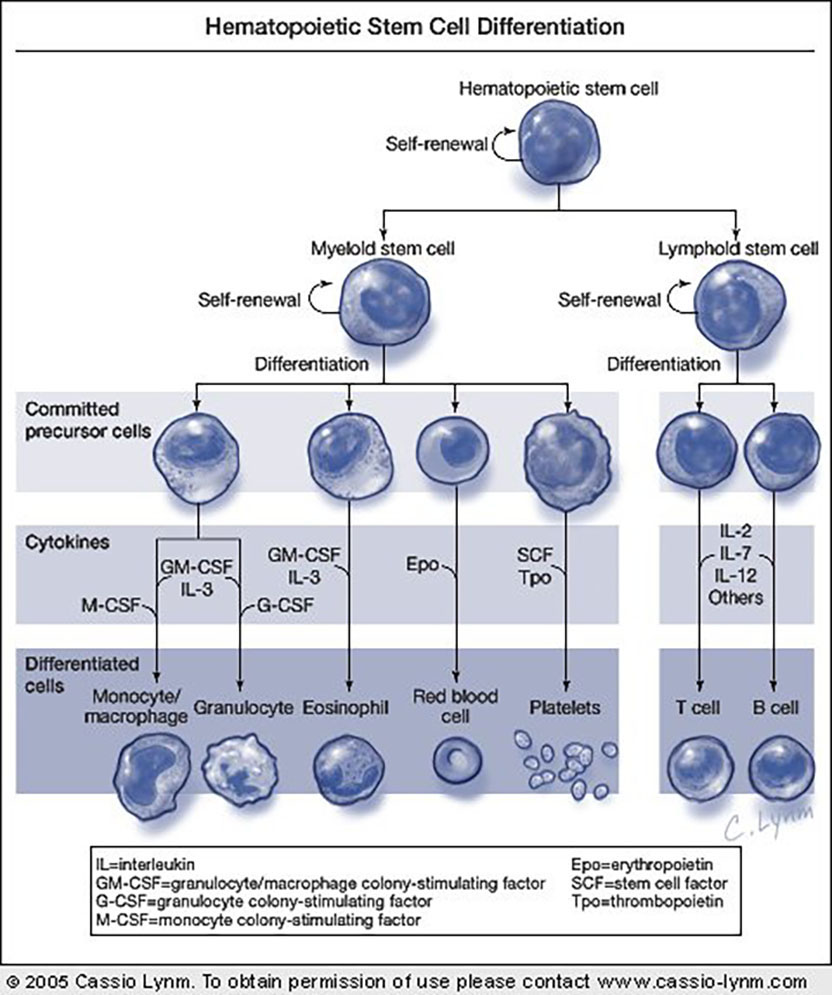
From stem to stern. This simplified diagram of hematopoiesis illustrates the two main properties of stem cells — self-renewal and differentiation — defined by Till and McCulloch. Each stem cell can either replace itself or can begin the path toward specialization, becoming a committed precursor cell. Precursor cells are converted to differentiated cells through the action of cytokines, some of which are shown. Some details of this scheme are still controversial.
Randomness and genetic programs
A particular feature of the results struck the scientists, however. Although nearly all of the spleen nodules contained new colony-forming units, some had many and some had few or none.
They repeated the experiment with these second-round cells to find out whether the colonies would breed true: Would a nodule from a colony that had produced many nodules contain many colony-forming cells? The results they obtained indicated that the number of new colonies produced wasn’t genetically programmed; instead it was random. Borrowing from the field of cosmic radiation, Till worked out a theory in which chance determined a stem cell’s fate — whether it would begin to differentiate or instead divide to generate two new stem cells. Till tested this model of spleen-colony growth by computer simulation and the results agreed with the experimental observations. The theory remains strong today, more than four decades after its conception.
The researchers next homed in on molecules in the stem cells and the blood-forming environment that play crucial roles in stem-cell function. Elizabeth Russell and Seldon Bernstein, of the Jackson Laboratory in Bar Harbor, Maine, studied a particular strain of mouse that was anemic and exceptionally susceptible to radiation. The animals’ anemia could be cured by injection of cells from mice that carried a regular version of the so-called “W” gene. McCulloch, Till, and Siminovitch showed that these genetically normal animals were donating colony-forming cells to their anemic siblings. Furthermore, they found that bone marrow from the anemic mice didn’t form colonies when injected into genetically intact but irradiated mice. The anemic mice therefore carried a genetically encoded defect in their blood-forming stem cells.
A different strain of mouse — with flaws in the “Sl” gene — seemed very similar to the “W” mice, at least on the surface. These animals were also anemic and unusually radiation sensitive. So Till, McCulloch, and Siminovitch performed analogous experiments on them, expecting similar results. The results surprised them. Marrow from these mice behaved normally when injected into irradiated recipients. However, marrow from genetically normal mice didn’t cure their anemia. These observations suggested that, rather than carrying defects in the stem cells themselves, the anemic “Sl” mice failed to support stem-cell growth. The results established the importance of the tissue environment in promoting normal stem cell duplication and specialization. Together, the work on “Sl” and “W” opened the door to the study of genetic regulation of stem-cell formation in mice, setting the stage for finding hematopoietic cytokines — proteins made by cells that affect the behavior of other cells — and their cellular receptors.
McCulloch and Till set a high standard for work on cell progenitors, and their findings strongly supported the hypothesis that cells with the capacity to self-renew, divide, and differentiate along many lineages existed and were available for rigorous analysis in adult animals. This finding paved the way for current attempts to physically isolate such cells, study their characteristics, and develop them for medical use. It also encouraged the pursuit of other types of stem cells, including embryonic stem cells. Like the stem cells they discovered, Till and McCulloch’s work has differentiated and matured in many directions.
by Evelyn Strauss
Key publications of Ernest McCulloch and James Till
McCulloch, E. and Till, J.E. (1960). The radiation sensitivity of normal mouse bone marrow cells, determined by quantitative marrow transplantation into irradiated mice. Rad. Res. 13, 115–125.
Till, J.E. and McCulloch, E.A. (1961). A direct measurement of the radiation sensitivity of normal mouse bone marrow cells. Rad. Res. 14, 213–222.
Becker, A.J., McCulloch, E.A., and Till, J.E. (1963). Cytological demonstration of the clonal nature of spleen colonies derived from transplanted mouse marrow cells. Nature. 197, 452.
Siminovitch, L., McCulloch, E.A., and Till, J.E. (1963). The distribution of colony-forming cells among spleen colonies. J. Cell. Comp. Physio. 62, 327.
Till, J.E., McCulloch, E.A., and Siminovitch, L. (1964). A stochastic model of stem cell proliferation, based on the growth of spleen colony-forming cells. Proc. Natl. Acad. Sci. USA. 51, 29–36.
McCulloch, E.A., Siminovitch, L., and Till, J.E. (1964). Spleen colony formation in anemic mice of genotype W/Wv. Science. 144, 844–846.
McCulloch, E.A., Siminovitch, L., Till, J.E., Russell, E.S., and Bernstein, S.E. (1965). The cellular basis of the genetically determined hemopoietic defect in anemic mice of genotype Sl/Sld. Blood. 26, 399–410.
Till, J.E. and McCulloch E.A. (1980). Hemopoietic stem cell differentiation. Biochim. Biophys. Acta. 605, 431–459.


 To begin to explain what today's Basic Science Lasker Award winners, Ernest McCullough and James Till, accomplished, let me point out that the reason we are all enjoying this luncheon is that we have blood cells — red blood cells that carry oxygen around, white blood cells that protect us from germs and platelets that keep us from bleeding. We constantly produce these cells, and we make trillions of them every day.
To begin to explain what today's Basic Science Lasker Award winners, Ernest McCullough and James Till, accomplished, let me point out that the reason we are all enjoying this luncheon is that we have blood cells — red blood cells that carry oxygen around, white blood cells that protect us from germs and platelets that keep us from bleeding. We constantly produce these cells, and we make trillions of them every day.


